Case Study Question on Amines
Read the passage given below and answer the following questions:
Reductive alkylation is the term applied to the process of introducing alkyl groups into ammonia or a primary or secondary amine by means of an aldehyde or ketone in the presence of a reducing agent. The present discussion is limited to those reductive alkylations in which the reducing agent is hydrogen and a catalyst or “nascent” hydrogen, usually from a metal- acid combination; most of these reductive alkylations have been carried out with hydrogen and a catalyst. The principal variation excluded is that in which the reducing agent is formic acid or one of its derivatives; this modification is known as the Leuckart reaction. The process of reductive alkylation of ammonia consists in the addition of ammonia to a carbonyl compound and reduction of the addition compound or its dehydration product. The reaction usually is carried out in ethanol solution when the reduction is to be effected catalytically
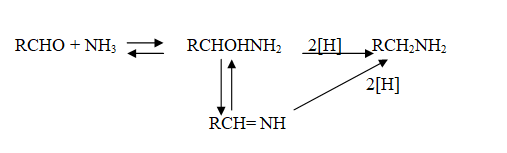
Since the primary amine is formed in the presence of the aldehyde it may react in the same way as ammonia, yielding an addition compound, a Schiff’s base (RCH= NCH2R) and finally, a secondary amine. Similarly, the primary amine may react with the imine, forming an addition product which also is reduced to a secondary amine. Finally, the secondary amine may react with either the aldehyde or the amine to give products which are reduced to tertiary amines.
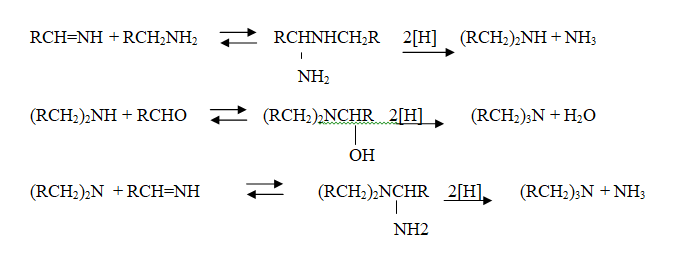
Similar reactions may occur when the carbonyl compound employed is a ketone.
(source: Emerson, W. S. (2011). The Preparation of Amines by Reductive Alkylation. Organic Reactions, 174–255. doi:10.1002/0471264180.or004.03 )
Q1. Ethanal on reaction with ammonia forms an imine (X) which on reaction with nascent hydrogen gives (Y). Identify ‘X’ and ‘Y’.
- X is CH3CH=NH and Y is CH3NH2
- X is CH3CHOHNH2 and Y is CH3CH2NH2
- X is CH3CHOHNH2 and Y is CH3NH2
- X is CH3CH=NH and Y is CH3CH2NH2
Q2. Acetaldehyde is reacted with ammonia followed by reduction in presence of hydrogen as a catalyst. The primary amine so formed further reacts with acetaldehyde. The Schiff’s base formed during the reaction is:
- CH3CH=NHCH3
- CH3CH=NHCH2CH3
- CH3=NHCH2CH3
- CH3CH2CH=NHCH3
Q3. The reaction of ammonia and its derivatives with aldehydes is called:
- Nucleophilic substitution reaction
- Electrophilic substitution reaction
- Nucleophilic addition reaction
- Electrophilic addition reaction
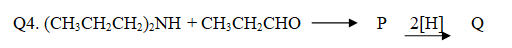
The compound Q is:
- (CH3CH2CH2)3N
- (CH3CH2CH2)2N(CH2CH3)
- (CH3CH2)3N
- (CH3CH2)2NH
Q5. Reductive alkylation of ammonia by means of an aldehyde in presence of hydrogen as reducing agents results in formation of:
- Primary amines
- Secondary amines
- Tertiary amines
- Mixture of all three amines

Pingback: Case Study Question 2 on Amines – Chapter 13 CBSE Class 12 Chemistry - Neutron Classes
Pingback: Case Study Question 3 on Amines – Chapter 13 CBSE Class 12 Chemistry – Neutron Classes - Neutron Classes